Check our 3-Way Catalytic Converters designed especially for gasoline and gaseous LSI and SSI engines
How Can We Reduce LPG Emissions?
Several approaches to control emissions from LPG engines operating in confined spaces are illustrated in the following chart. Usually a combination of available methods provides the optimum solution.
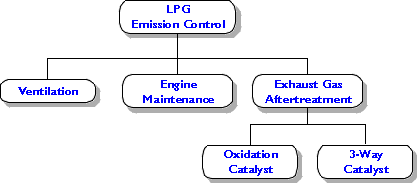 Figure 5. Methods of LPG Emission Control
Figure 5. Methods of LPG Emission Control
Ventilation is always required in confined work environments, no matter whether engines operate in the area or not. If engines do operate, higher ventilation rates should be provided. As a guideline, each LPG forklift truck with a 60 hp engine and not equipped with an emission control catalyst requires about 2.4 m3/s (5000 cfm) of ventilation air. If electrical heating is used, this number translates to over $500.00 per month of extra heating cost during winter (heating from 0 to 18°C, 8 hrs per day, 21 days per month, energy cost 0.06 $/kWh). Another disadvantage of ventilation as a means of emission control is its uneven distribution. It is very difficult to provide perfect air flow, mixing, and distribution throughout the ventilated area. Usually certain parts of the building obtain less ventilation than others and accumulate higher concentrations of CO and other pollutants. In particular, high CO concentrations are frequently observed in proximity of operating LPG vehicles. Sometimes LPG forklifts operate in areas with extremely poor ventilation, e.g., inside large transport trucks.
Engine tune-up and proper maintenance are invaluable tools to control carbon monoxide emissions from LPG engines. A regular maintenance program should be implemented which incorporates final engine tuning through carbon monoxide analysis of exhaust gases. A CO analyzer working on the infrared (IR) principle provides the highest accuracy and best reliability. Carbon monoxide concentrations in the exhaust gases should be always below 1%. On most engines the CO level can be kept below 0.5% through good engine tune-up.
Catalytic exhaust gas aftertreatment provides the final means of LPG emission control. Tailpipe carbon monoxide levels as low as 100 ppm can be achieved depending, however, on the CO concentrations in the raw exhaust. It is very important to realize that exhaust aftertreatment is not a substitute for good engine maintenance and tune-up. On the contrary, exhaust gas aftertreatment is complementary to engine maintenance and, in fact, requires itself that the engine is in good condition and no excessive raw emissions are observed. Catalytic conversion of high concentrations of carbon monoxide and hydrocarbons generates large quantities of heat. As a result the catalyst temperature would raise, leading to its overheating and premature loss of activity.
Oxidation catalyst systems are commonly used today for emission control of LPG forklifts and off-road vehicles. The oxidation catalyst, sometimes also called 2-way catalyst, is effective in reducing two major exhaust pollutants, i.e., carbon monoxide and hydrocarbons. Both are removed from the exhaust through oxidation to carbon dioxide and water vapor (Figure 6).
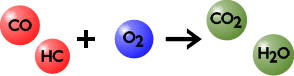 Figure 6. Chemistry in Oxidation Catalyst
Figure 6. Chemistry in Oxidation Catalyst
A newer catalyst technology is known as the 3-way catalyst. Three pollutants are simultaneously removed from the exhaust gas in the 3-way catalyst. They are carbon monoxide, hydrocarbons, and nitrogen oxides. The chemistry occurring in the 3-way catalyst is schematically illustrated in Figure 7.
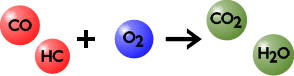 Figure 7. Chemistry in 3-Way Catalyst
Figure 7. Chemistry in 3-Way Catalyst
The fundamental reactions in the 3-way catalyst are between CO and HC on one side and NOx on the other. To achieve high conversions of all three pollutants, their concentrations must be at the stoichiometric ratio. It simply means that the amount of CO and HC should match the amount of NOx present in the system, as reflected in the exact equations of chemical reactions which occur on the catalyst. A precise electronic engine controller maintains that ratio on a real engine, as happens in today’s automobiles. 3-way catalyst systems are available from Nett Technologies and other vendors, while oxidation systems are now less frequently used for LPG emission control.
Nett Technologies’ 3-Way Catalytic Converters

Request A Quote
Photo Gallery

