Check our M-Series (Metallic) and D-Series (Ceramic) catalytic converters designed especially for diesel engines…
What Are Diesel Emissions?
Diesel engines convert the chemical energy contained in the fuel into mechanical power. Diesel fuel is injected under pressure into the engine cylinder where it mixes with air and where the combustion occurs. The exhaust gases which are discharged from the engine contain several constituents that are harmful to human health and to the environment.
Table 1 lists typical output ranges of the basic toxic material in diesel fumes. The lower values can be found in new, clean diesel engines, while the higher values are characteristic for older equipment.
| CO | HC | DPM | NOx | SO2 |
|---|---|---|---|---|
| vppm | vppm | g/m3 | vppm | vppm |
| 5-1,500 | 20-400 | 0.1-0.25 | 50-2,500 | 10-150 |
Carbon monoxide (CO), hydrocarbons (HC), and aldehydes are generated in the exhaust as the result of incomplete combustion of fuel. A significant portion of exhaust hydrocarbons is also derived from the engine lube oil. When engines operate in enclosed spaces, such as underground mines, buildings under construction, tunnels or warehouses, carbon monoxide can accumulate in the ambient atmosphere and cause headaches, dizziness and lethargy. Under the same conditions, hydrocarbons and aldehydes cause eye irritation and choking sensations. Hydrocarbons and aldehydes are major contributors to the characteristic diesel smell. Hydrocarbons also have a negative environmental effect, being an important component of smog.
Nitrogen oxides (NOx) are generated from nitrogen and oxygen under the high pressure and temperature conditions in the engine cylinder. NOx consist mostly of nitric oxide (NO) and a small fraction of nitrogen dioxide (NO2). Nitrogen dioxide is very toxic. NOx emissions are also a serious environmental concern because of their role in the smog formation.
Sulfur dioxide (SO2) is generated from the sulfur present in diesel fuel. The concentration of SO2 in the exhaust gas depends on the sulfur content of the fuel. Low sulfur fuels of less than 0.05% sulfur are being introduced for most diesel engine applications throughout the USA and Canada. Sulfur dioxide is a colorless toxic gas with a characteristic, irritating odor. Oxidation of sulfur dioxide produces sulfur trioxide which is the precursor of sulfuric acid which, in turn, is responsible for the sulfate particulate matter emissions. Sulfur oxides have a profound impact on environment being the major cause of acid rains.
Diesel particulate matter (DPM), as defined by the EPA regulations and sampling procedures, is a complex aggregate of solid and liquid material. Its origin is carbonaceous particles generated in the engine cylinder during combustion. The primary carbon particles form larger agglomerates and combine with several other, both organic and inorganic, components of diesel exhaust. Generally, DPM is divided into three basic fractions (Figure 1):
- Solids - dry carbon particles, commonly known as soot,
- SOF - heavy hydrocarbons adsorbed and condensed on the carbon particles, called Soluble Organic Fraction,
- SO4 - sulfate fraction, hydrated sulfuric acid.
The actual composition of DPM will depend on the particular engine and its load and speed conditions. "Wet" particulates can contain up to 60% of the hydrocarbon fraction (SOF), while "dry" particulates are comprised mostly of dry carbon. The amount of sulfates is directly related to the sulfur contents of the diesel fuel.
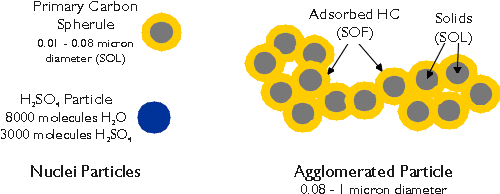
Figure 1. Schematic Composition of Diesel Particulate Matter
Diesel particulates are very fine. The primary (nuclei) carbon particles have a diameter of 0.01 - 0.08 micron, while the agglomerated particles diameter is in the 0.08 to 1 micron range. As such, diesel particulate matter is almost totally respirable and has a significant health impact on humans. It has been classified by several government agencies as either "human carcinogen" or "probable human carcinogen". It is also known to increase the risk of heart and respiratory diseases.
Polynuclear Aromatic Hydrocarbons (PAH) are hydrocarbons containing two or more benzene rings. Many compounds in this class are known human carcinogens. PAHs in the exhaust gas are split between gas and particulate phase. The most harmful compounds of four and five rings are present in the organic fraction of DPM (SOF).
Nett Technologies’ Diesel Oxidation Catalysts (DOC)

Nett Technologies’ Diesel Particulate Filters (DPF)
Manufactured using either Cordierite or Silicon Carbide substrates, DPFs are an effective solution to Diesel…

Nett Technologies’ Selective Catalytic Reduction (SCR) Systems
Designed to control the emissions of Nitrogen Oxides (NOx) from medium to heavy duty diesel…

Request A Quote
Photo Gallery

